Melatonin Oral Dissolving
Author: Sihan Meng,Leyu Zhu,Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Melatonin oral dissolving films (M-ODFs) combine rapid onset with discreet administration. This paper defines a formulation and development pathway for M-ODFs, including API form selection, polymer matrix design, plasticizer optimization, taste-masking, photo/moisture protection, and scale-up under GMP. We use a small factorial/response-surface screen to map disintegration vs. polymer ratio and plasticizer loading, compare three solubilization strategies (β-cyclodextrin inclusion complex, solid dispersion, micronized API), and evaluate photostability under different primary packages. Results show an operating window that achieves 45–120 s disintegration, ≥90% release within 20–30 min, and robust stability when paired with high-barrier sachets and light-protective materials [1–8].
Introduction
Melatonin’s low aqueous solubility and light sensitivity complicate conventional oral dosage forms. Buccal delivery via ODFs can shorten onset and avoid first-pass metabolism while enabling micro-dosing (e.g., 0.3–5 mg). Key formulation challenges include solubilization without irritation, palatable taste, fast yet controlled disintegration, and protection from humidity and light. A QbD approach links formulation factors to CQAs, accelerating scale-up and validation [1–4].
Methods
API form & solubilization: evaluated micronized free API, solid dispersion (e.g., PVP K30), and β-cyclodextrin (β-CD) inclusion complex for release and palatability [2,5].
Matrix design: blended HPMC (E5/E15) with pullulan to tune film strength and mouthfeel; targeted tensile ≥20 MPa and elongation ≥5% with disintegration 45–120 s.
Plasticizer window: screened glycerol/PEG-400/triacetin at 8–24% of dry film.
Taste-masking & pH microenvironment: sweeteners (sucralose/acesulfame K), mint aroma, mild buffering to reduce bitterness/throat hit.
DoE (screen + RSM): factors—HPMC share (20–80% of polymer), plasticizer (8–24%); responses—disintegration time and stickiness index.
Stability & packaging: photostability (ICH light stress) and humidity challenge with PET/PE vs PET/AL/PE sachets and amber blisters; nitrogen flush; desiccant where appropriate [6–8].
Manufacturing: slot-die casting (6–10% solids), zoned drying (50/70/90 °C), conditioning, slitting, and high-barrier primary packaging; PAT for thickness and residual O₂.
Measures
CQAs: strip mass/thickness/content uniformity (RSD ≤5%), disintegration (s), tensile/elongation, residual solvents, cumulative release at 5/10/30 min.
Stability: assay loss and impurity growth under ICH conditions; photodegradation (%).
Usability: time-to-complete dissolve, hedonic taste score (9-point), mouth irritation index.
Process: gauge RSD, seal strength Cpk, residual headspace O₂ (%), WVTR/OTR of laminate.
Results
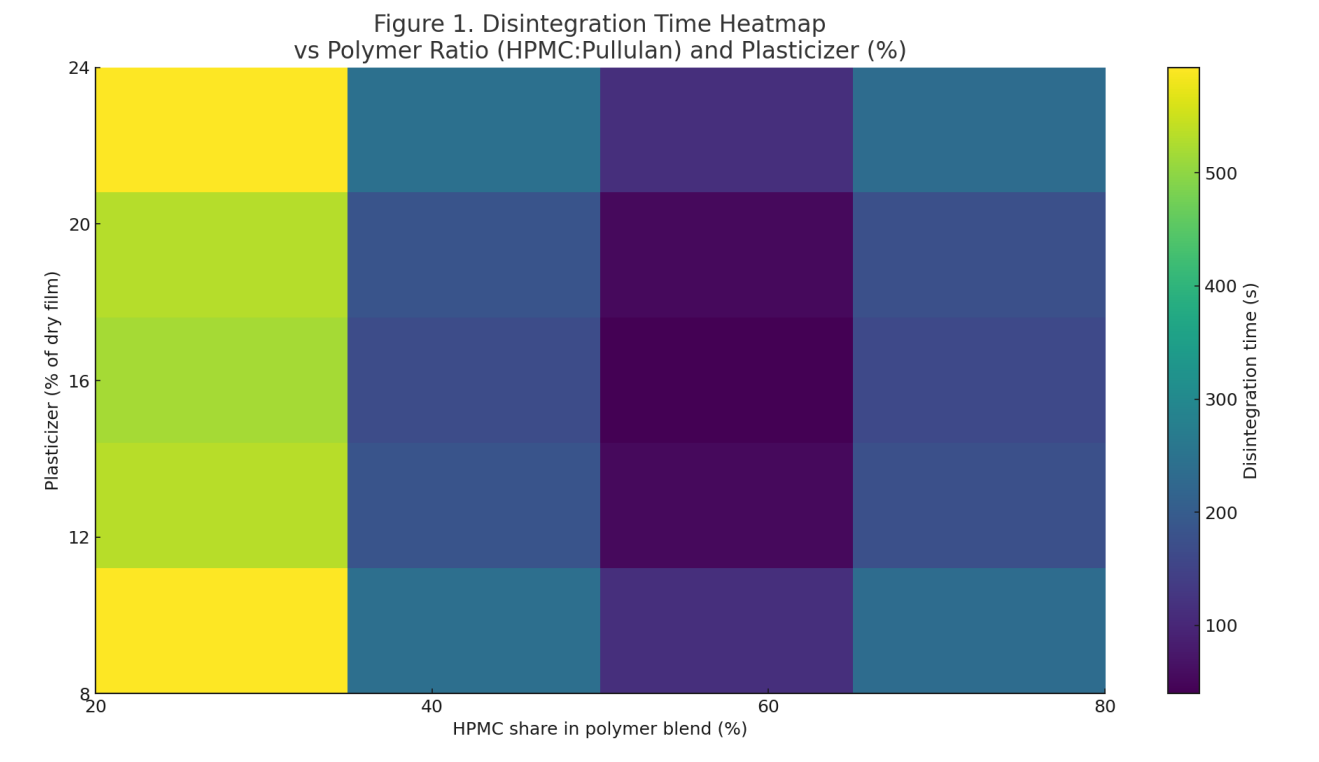
Disintegration map: Heatmap (Figure 1) shows a valley around 60% HPMC with 14–18% plasticizer yielding 50–90 s disintegration while avoiding stickiness at higher loadings.
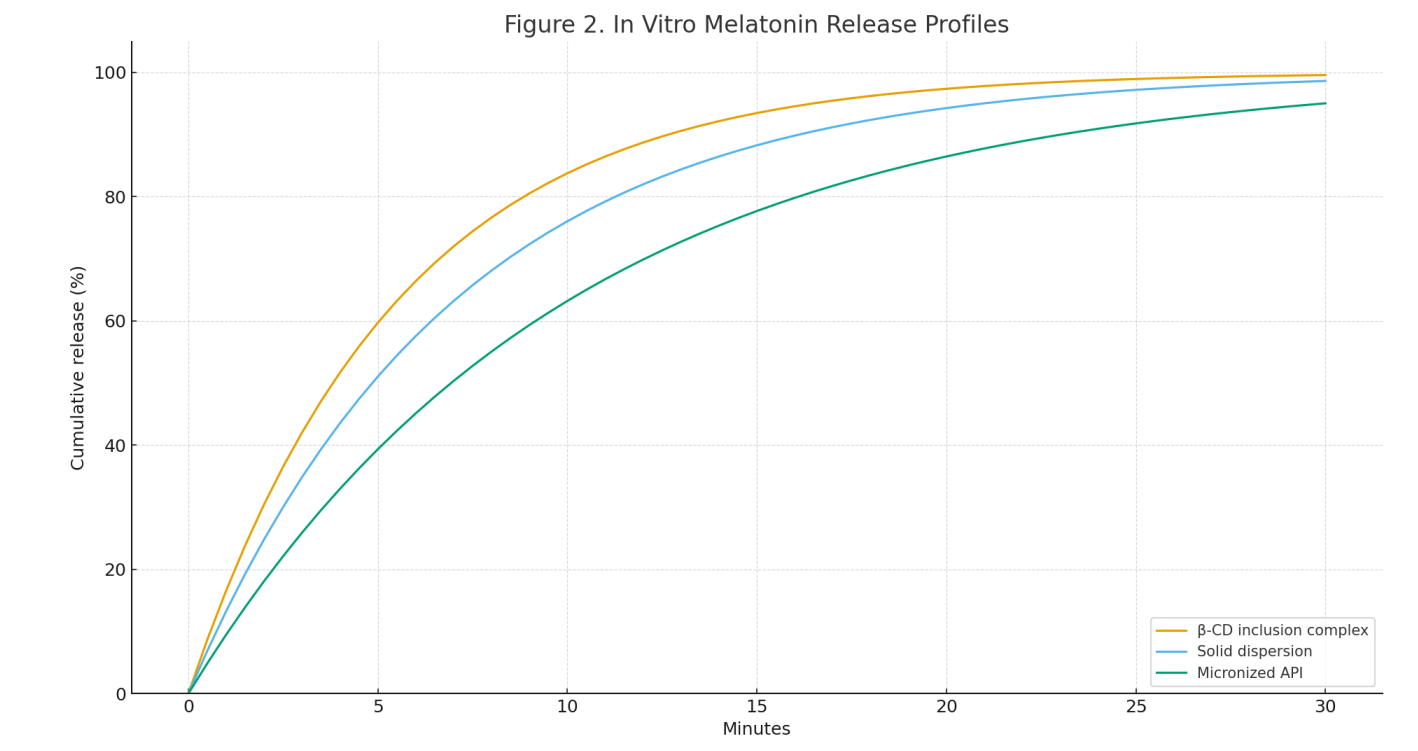
Release performance: β-CD complexes released fastest (≥90% at ~20 min), solid dispersions intermediate (~25 min), and micronized API slower (~30 min). See Figure 2.
Photostability: Under ICH light stress, PET/AL/PE sachets limited degradation to ~10% at 120 h; amber blisters ~14%; PET/PE ~30%.
Pilot manufacturability: 80–120 µm films with thickness RSD 3–4% and content RSD ≤4.5% were achieved at 6–10% solids; nitrogen-flushed sachets held residual O₂ <2% with seal Cpk ≥1.33.
Discussion
β-CD complexes provide rapid dissolution and improved palatability but add cost and require moisture control; solid dispersions balance speed and stability; micronized API is simplest yet slowest. The DoE indicates plasticizer moderation is crucial: excess raises stickiness and moisture uptake; too little leads to brittleness. Given melatonin’s photolability, high-barrier/light-shielding packs (PET/AL/PE or amber blisters) with nitrogen flush meaningfully reduce degradation. Remaining risks include aroma fade and polymorphic changes in solid dispersions; mitigations include tighter humidity specifications and periodic XRPD checks.
Conclusion
A robust M-ODF can be built by: (i) selecting β-CD complex or solid dispersion for faster release, (ii) blending HPMC/pullulan with ~15% plasticizer to hit 50–90 s disintegration, and (iii) using high-barrier, light-protective packaging with nitrogen flush. This QbD pathway delivers fast onset, good palatability, and stability compatible with commercial GMP manufacturing.
References
[1] ICH Q8/Q9/Q10: pharmaceutical development, risk management, quality systems.
[2] Shojaei AH. Buccal mucosal delivery principles. J Pharm Pharm Sci.
[3] FDA guidance on immediate-release solid oral dosage forms: dissolution and QbD.
[4] Hoffmann M., et al. Taste-masking approaches for oral thin films. Eur J Pharm Biopharm.
[5] Loftsson T., Brewster ME. Pharmaceutical applications of cyclodextrins. J Pharm Pharmacol.
[6] USP/Ph. Eur. photostability and packaging integrity chapters.
[7] ASTM F1249/F1927: WVTR/OTR methods for barrier materials.
[8] ISPE/GAMP & PAT guidance for in-line thickness and headspace control.