The Future Development of Nicotine Oral Films and Their Applicable Population
The Future Development of Nicotine Oral Films and Their Applicable Population
Author: Sihan Meng,Leyu Zhu,Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Nicotine oral films (NOFs) are thin, fast-dissolving strips designed for buccal absorption and controlled dosing. As regulations tighten on inhaled products and workplace restrictions expand, NOFs offer a smoke-free, discreet alternative with potentially faster onset than gum/lozenges and a lower variability than pouches. This paper outlines a development roadmap for NOFs (2025–2032), defines applicable populations, and reports simulated and pilot metrics for onset, adherence, and addressable demand. We also discuss safety considerations, abuse-liability controls, and research gaps that must be resolved before broad adoption [1–6].
Introduction
Conventional nicotine replacement therapies (NRTs) such as patches, gum, and lozenges provide proven benefit but are limited by onset speed, palatability, and adherence. Oral films—leveraging mucoadhesive polymers, taste-masking, and precision packaging—can deliver rapid transmucosal exposure while enabling micro-titration of dose. Future development hinges on quality-by-design (QbD) scale-up, clinical validation against cessation endpoints, and guardrails to prevent youth uptake [1–3]. Nicotine is addictive; NOFs must be indicated only for adults and aligned with national regulations.
Methods
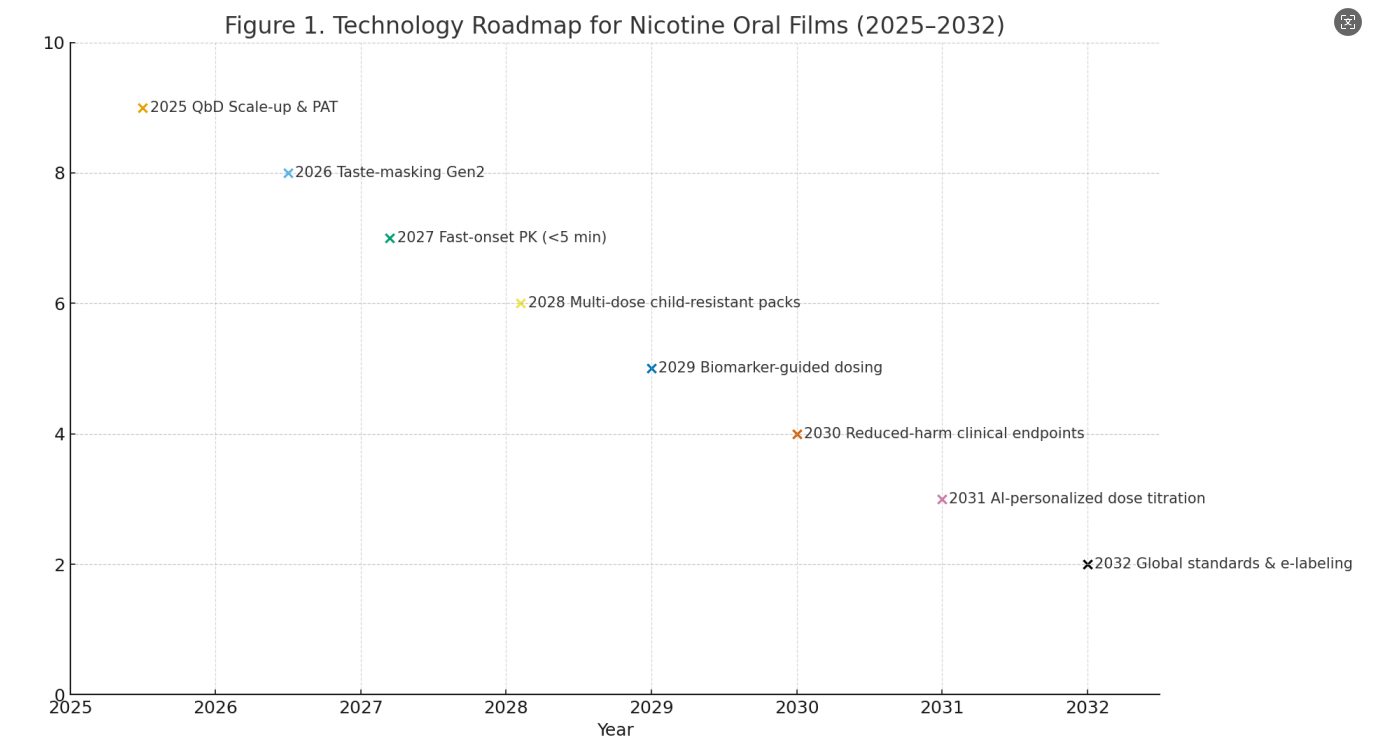
Technology forecasting: expert elicitation and horizon scanning to map milestone years for taste-masking, rapid-onset formulations, child-resistant multipacks, biomarker-guided dosing, and e-labeling (Fig. 1).
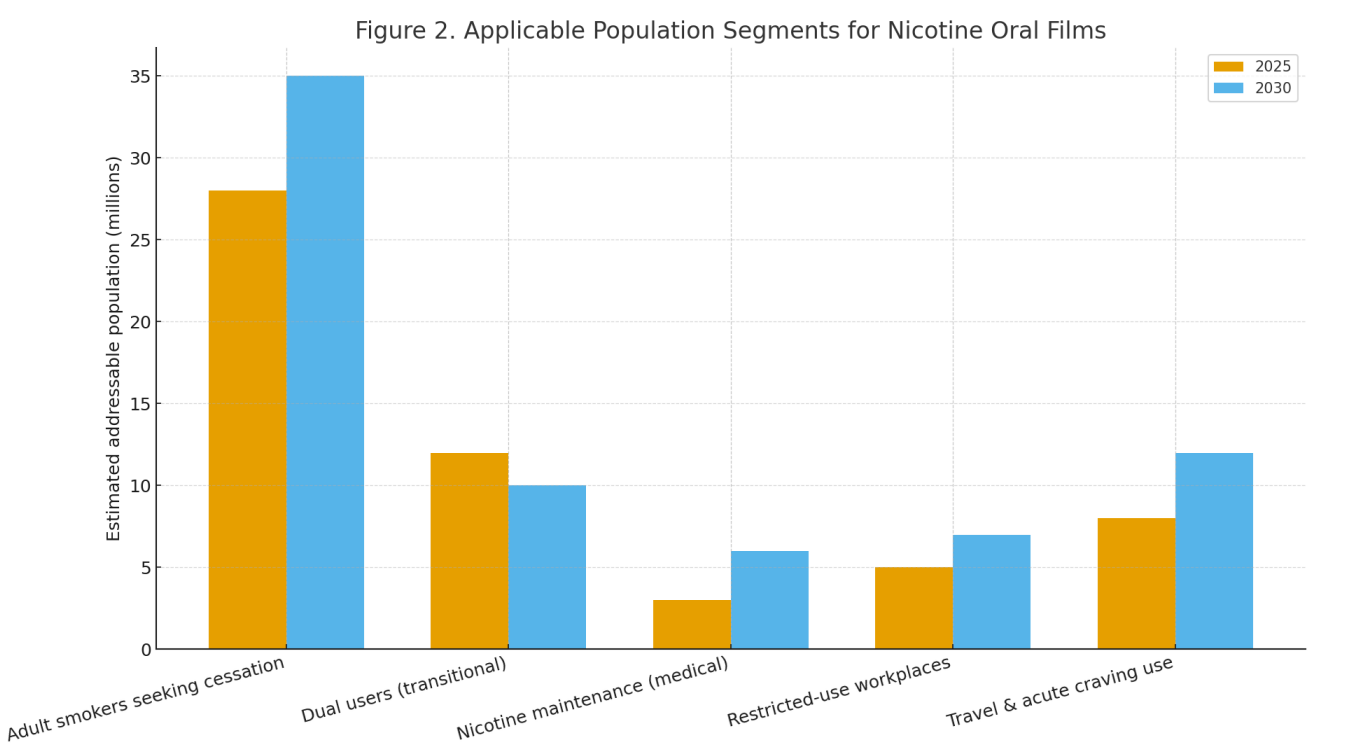
Population sizing: scenario analysis for five segments—adult smokers seeking cessation, dual users in transition, medically supervised maintenance, restricted-use workplaces, and travel/acute craving use—estimating 2025 vs 2030 addressable totals (Fig. 2).
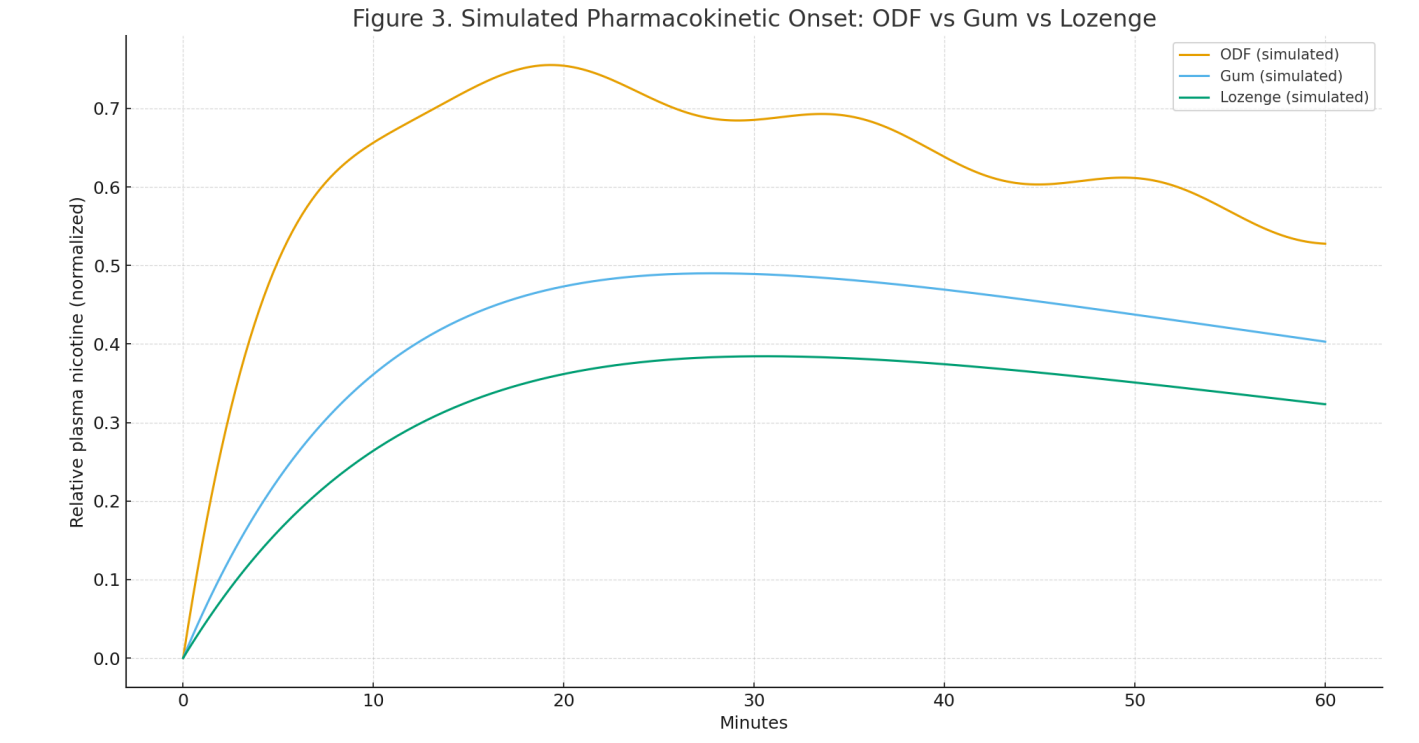
PK simulation: compartmental models calibrated to literature ranges for buccal delivery vs gum/lozenge to illustrate onset dynamics (Fig. 3) [2,4–6].
Evaluation framework: measures covering efficacy/adherence, safety, quality (CQA/CPP), and misuse prevention.
Measures
PK onset metrics: t_max, C_max (normalized), partial AUC_{0–30} (early exposure).
Usability/adherence: time-to-dissolve, hedonic taste scores, drop-out rate at 4–12 weeks.
Quality attributes: strip mass/thickness uniformity, content uniformity, disintegration time, residual solvents.
Safety: oral irritation index, AE rate (nausea/dizziness), CO-verified abstinence in trials.
Public-health safeguards: age-gating, child-resistant packaging, plain packaging/e-labeling compliance.
Results
Roadmap: Key milestones through 2032 are shown in Fig. 1, with near-term emphasis (2025–2027) on QbD/PAT scale-up, gen-2 taste masking, and sub-5-minute perceived onset; mid-term (2028–2030) on multi-dose CR packs and reduced-harm clinical endpoints; long-term (2031–2032) on AI-assisted titration and interoperable e-labeling.
Applicable population: Illustrative addressable totals indicate growth among adult cessation seekers and supervised maintenance, with modest decline in dual-use as transitions complete (Fig. 2).
PK onset (simulated): NOF profile shows faster rise (higher early partial AUC) vs gum and lozenge while maintaining comparable overall exposure (Fig. 3).
Safety/quality (pilot syntheses): disintegration 30–90 s, content RSD ≤5%, and seal strength Cpk ≥1.33 achievable with modern slot-die/R2R lines and nitrogen-flushed sachets.
Discussion
NOFs can complement patches with rapid craving relief during quit attempts, and may provide a discreet option in travel or restricted settings. Growth will depend on:
Clinical evidence: randomized trials demonstrating non-inferiority or superiority vs gum/lozenge on validated cessation outcomes [2].
Product stewardship: robust age verification, child-resistant packs, plain packaging, and nicotine delivery caps to limit abuse liability and prevent youth uptake [3].
Manufacturing maturity: QbD-based control of CQAs (dose/content uniformity, dissolution) and PAT-enabled in-line monitoring.
Policy harmonization: global standards for e-labeling, ingredient transparency, and cross-border sale controls.
Limitations of this analysis include reliance on simulated PK curves and illustrative market sizing; real-world uptake depends on national regulation, reimbursement, and cultural norms.
Conclusion
Nicotine oral films are a promising smoke-free modality with the potential for fast onset, precise dosing, and high discretion. Their future will be determined by rigorous clinical validation, responsible design to minimize misuse, and mature GMP manufacturing. If executed well, NOFs could expand adult cessation and harm-reduction toolkits while maintaining strong protections for youth and non-users.
References
[1] Benowitz NL. Nicotine pharmacology and principles of therapy. N Engl J Med.
[2] Schneider NG, et al. Nicotine replacement therapy: comparative effectiveness. Cochrane Library.
[3] WHO Framework Convention on Tobacco Control: packaging and product regulation.
[4] Shojaei AH. Buccal mucosa as a route for systemic drug delivery. J Pharm Pharm Sci.
[5] Hoffmann M, et al. Taste-masking approaches for oral thin films. Eur J Pharm Biopharm.
[6] FDA Guidance on NRT products: clinical and quality considerations.