End-to-End Manufacturing Process for Oral Dissolving Films (ODFs)
Author: Pengcheng(Frank) Shi,Leyu Zhu,Sihan Meng
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Oral dissolving films (ODFs) are thin polymeric matrices designed to disintegrate rapidly in the oral cavity and deliver active substances without water. This paper delineates an end-to-end, GMP-compliant manufacturing workflow from pre-formulation through primary/secondary packaging. It integrates quality-by-design (QbD) principles, process analytical technology (PAT), equipment selection, critical process parameters (CPPs), and critical quality attributes (CQAs). Emphasis is placed on solution preparation, filtration and deaeration, precision coating, staged drying and curing, web handling, slitting and singulation, and high-barrier unit packaging. Scale-up, validation (IQ/OQ/PQ), cleaning, data integrity, and EHS/ATEX controls for solvent handling are discussed. A risk-based control strategy is proposed to ensure dose uniformity, rapid disintegration (≈10–30 s), mechanical robustness, low residual solvents, and long-term stability.¹²³⁴
Keywords: oral dissolving films; roll-to-roll; QbD; PAT; slot-die coating; GMP; CQAs
1. Introduction
ODFs have matured from lab curiosity to a robust oral transmucosal platform for pharmaceutical and nutraceutical products. Their continuous roll-to-roll (R2R) manufacturability, fast onset, and patient-centric use drive adoption. However, small unit dose, thin geometries, and moisture/oxygen sensitivity demand precise process control and barrier packaging. A QbD framework linking CQAs to material attributes and CPPs is essential for reproducibility and regulatory acceptance.¹²
2. Materials and Pre-Formulation
Typical matrices use hydrophilic film-formers (e.g., HPMC, PVA, pullulan), plasticizers (glycerol, propylene glycol, low-MW PEG), sweeteners/flavors, buffering agents, and—when required—solubilizers or lipidic dispersions (e.g., nanoemulsions, cyclodextrin complexes) for poorly soluble actives.³ Compatibility screens (DSC/FT-IR), oxidation/light sensitivity checks, and accelerated stability guide excipient selection and oxygen/moisture specifications. Dose range, target disintegration, and mouthfeel inform solids content and viscosity windows for coatability.¹³
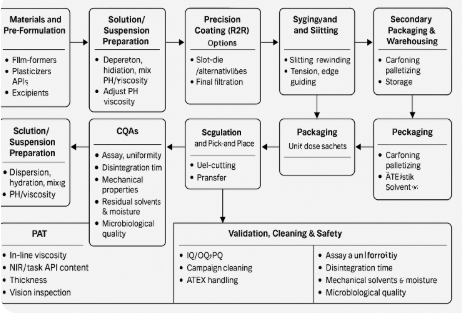
3. Process Flow (Unit Operations)
3.1 Solution/Suspension Preparation
Steps: Premix solvent (water and/or approved organics), disperse polymer under controlled shear, age for full hydration, add API and minor excipients, adjust pH and viscosity, then polish-filter.
CPPs: solids content, viscosity (rheology curve), temperature, pH, shear history, particle size for suspensions.
Controls: jacketed vessels with sanitary agitation, in-line filters (0.45–5 µm), nitrogen blanketing for oxygen-sensitive APIs, and explosion-protected equipment if flammable solvents are used.²⁴
3.2 Deaeration and Filtration
Vacuum or membrane deaerators remove entrained air to prevent pinholes and thickness chatter; final filtration (e.g., 20–50 µm bag + 5 µm polish) ensures nozzle cleanliness for precision coaters.³
3.3 Precision Coating (R2R)
Options: slot-die (preferred for uniformity), knife-over-roll, comma, gravure, or curtain coating depending on viscosity and target coat weight.
CPPs: wet gap/flow rate (slot-die), line speed, web tension, edge bead control, substrate selection and surface energy (e.g., PET release liner, corona-treated).
PAT: in-line beta/X-ray or optical gauges for wet-film thickness, vision systems for streaks/defects, and tension load-cells across zones.¹²³
3.4 Drying and Curing
Staged Drying: multi-zone convection or IR ovens with controlled exhaust and LEL monitoring; early zones focus on skin-free solvent removal, mid-zones on bulk drying, final zones on residual solvent and moisture specification.
CPPs: zone temperatures, air velocity, solvent partial pressure, web residence time, and dew point; target residual solvent and moisture tailored to API and matrix.
Curing/Conditioning: post-dry equilibration (e.g., 24–72 h at controlled RH) can relieve internal stress, stabilize dimensions, and set disintegration behavior.²⁴
3.5 Web Handling, Slitting, and Rewinding
CPPs: longitudinal/cross-web tension, nip pressure, lay-on rollers, anti-static bars.
Controls: edge guiding and closed-loop tension prevent wrinkle and gauge bands; slitter geometry (shear/razor/crush) chosen to minimize burrs and particulates.³
3.6 Singulation: Die-Cutting & Pick-and-Place
Rotary or flatbed die-cutting defines unit size (e.g., 20–40 mm squares). Vacuum pick-and-place transfers pieces to primary packages.
CPPs: die clearance, registration (color-mark or camera), cut depth, tool wear; particulate generation must meet limits for oral non-sterile forms.¹
3.7 Primary Packaging (Unit Dose)
High-barrier sachets (e.g., PET/Al/PE or foil/PE) or blister-foil combinations protect against moisture/oxygen and light.
CPPs: sealing temperature/pressure/dwell, sachet headspace and oxygen level, leak integrity (dye-ingress or vacuum decay).
Controls: in-line checkweighers or vision-based piece-presence, and serialization/traceability if required.²⁴
3.8 Secondary Packaging & Warehousing
Cartoning with desiccant strategy, palletization, and cold-chain if demanded by API stability. Storage conditions typically maintain low water activity and controlled temperature, with humidity-logging for trending.²
4. Critical Quality Attributes (CQAs) and Specifications
Assay & Content Uniformity: per-piece accuracy and RSD; small unit doses demand strict metering and homogeneity control.¹²
Disintegration Time: commonly targeted at ~10–30 s; correlates with polymer grade, plasticizer level, porosity, and residual moisture.³
Mechanical Properties: tensile strength/elongation and tear for handling; curl/flatness for packability.
Thickness and Coat Weight: tight tolerances for dose accuracy and mouthfeel.
Residual Solvents & Moisture: meet ICH Q3C and product-specific limits.²
Microbiological Quality: oral non-sterile limits; bioburden control via low aw, preservatives if justified, and hygienic design.
Organoleptics & Surface pH: taste, mouthfeel, and pH compatibility with oral mucosa.¹
5. QbD and Control Strategy
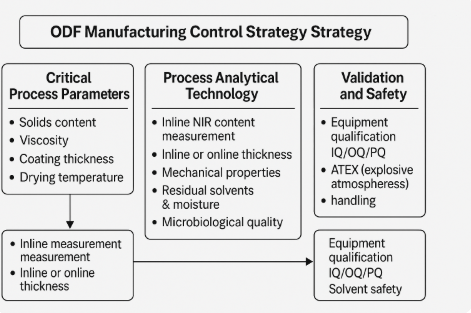
Using ICH Q8/Q9/Q10, map material attributes (polymer molecular weight, plasticizer %, API PSD) and CPPs (coat weight, oven profile, web tension) to CQAs via risk assessment (FMEA) and designed experiments (DoE). Define a design space for coat weight × line speed × drying profile that preserves dose uniformity, disintegration, and residual-solvent limits. Implement feedback/forward controls: viscosity-to-flow models for slot-die setpoints, tension cascades, and moisture-driven cure timers.¹²
6. Process Analytical Technology (PAT)
Recommended PAT includes: in-line viscosity (torsional/ultrasonic), NIR/ Raman to monitor solids and API content in the wet film, beta/X-ray gauges for thickness, machine vision for defects/registration, and end-of-line headspace oxygen. Electronic batch records (EBR) and data integrity controls (ALCOA+) support release by exception and deviation trending.²⁴
7. Scale-Up and Tech Transfer
Scale-up from lab (hand-cast or table-top coaters) to pilot and commercial lines requires dynamic similarity in coating window (Capillary number/flow regime) and matched drying trajectories (solvent flux vs. skinning). Conduct engineering runs to confirm tension distribution, oven balance, and edge-bead mitigation, then execute PPQ lots under the commercial control strategy.³⁴
8. Validation, Cleaning, and EHS
IQ/OQ/PQ: verify utilities (HVAC, compressed air quality), ATEX zoning, interlocks, and recipe management; qualify CPP ranges and PAT functionality.¹²
Cleaning Validation: worst-case APIs and flavors; swab/rinse recovery factors; campaign rules to prevent cross-contamination; riboflavin or tracer tests for coating heads and dies.³
EHS/ATEX: LEL monitoring, VOC capture/abatement, grounded web paths and static control, solvent storage per fire code, PPE and training.²⁴
9. Conclusions
Manufacturing ODFs at scale requires precise control of liquid handling, web coating, staged drying, and barrier packaging within a QbD framework. By coupling PAT with robust CPP governance and solvent/EHS controls, producers can consistently meet dose uniformity, rapid disintegration, stability, and patient-experience targets while satisfying global GMP expectations.¹²³⁴
References
¹ ICH Q8(R2)/Q9/Q10: Pharmaceutical Development, Quality Risk Management, and Pharmaceutical Quality System (foundational guidance for QbD and control strategy).
² FDA/EMA guidances on oral transmucosal/ODT quality considerations, data integrity, and process validation (IQ/OQ/PQ and continued verification).
³ Dixit R.P., Puthli S.P. “Oral Strip Technology: Overview and Future Potential.” J Control Release (overview of materials, coating, and packaging for films).
⁴ Technical monographs and ATEX/EHS best practices for solvent-based web coating lines, including PAT applications and LEL/VOC management in multi-zone dryers.