Oral Film Project: From Concept to Reality—A One-Stop Solution for Product Positioning, Formulation, Equipment, and Packaging
Oral Film Project: From Concept to Reality—A One-Stop Solution for Product Positioning, Formulation, Equipment, and Packaging
Author: Sihan Meng,Leyu Zhu,Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Oral Disintegrating Films (ODFs), also known as Oral Thin Films (OTFs), have evolved from niche pharmaceutical dosage forms into versatile platforms spanning pharmaceuticals, nutraceuticals, and functional consumer products. However, many ODF projects fail to reach commercialization due to fragmented development pathways—where product positioning, formulation design, manufacturing equipment, and packaging are handled in isolation. This paper presents a holistic, one-stop solution model for ODF project development, covering the full lifecycle from concept definition to commercial-ready production. By integrating market positioning, formulation engineering, equipment selection, and packaging design within a unified framework, this approach significantly reduces development risk, time-to-market, and cost. Practical workflows, evaluation measures, and industrial outcomes are discussed to provide a reproducible roadmap for successful ODF commercialization.
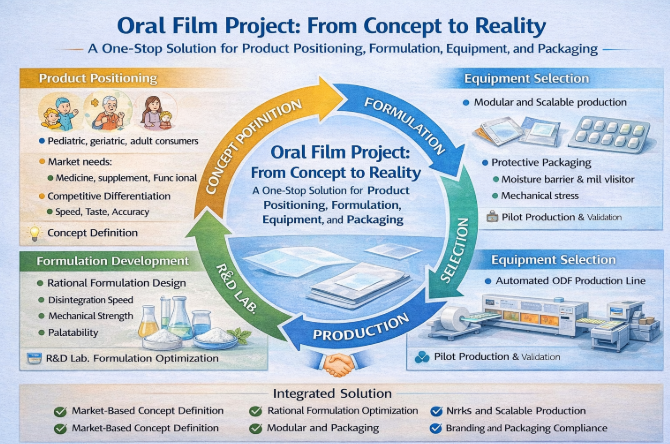
Introduction
The growing demand for fast-dissolving, portable, and patient-friendly oral dosage forms has accelerated interest in Oral Disintegrating Films worldwide [1]. Compared with tablets, capsules, and gummies, ODFs offer advantages such as rapid onset, precise dosing, ease of administration, and strong differentiation potential in crowded markets [2].
Despite these advantages, ODF development remains complex. Many projects focus narrowly on formulation while underestimating challenges related to scale-up, equipment compatibility, packaging stability, and regulatory alignment [3]. As a result, promising concepts often stall at pilot stage or encounter costly redesigns during commercialization. This paper proposes a one-stop ODF project solution that treats product positioning, formulation, equipment, and packaging as interdependent elements of a single system.
Concept and Product Positioning
Market-Oriented Concept Definition
An effective ODF project begins with clear product positioning rather than formulation alone. Key positioning dimensions include [4]:
Target population (pediatric, geriatric, adult consumers)
Use scenario (medical, daily supplement, on-the-go use)
Competitive differentiation (speed, taste, dosage accuracy, format novelty)
Regulatory pathway (drug, dietary supplement, functional product)
Early alignment of these factors defines technical boundaries for formulation and manufacturing.
Dosage Form Strategy
Positioning decisions determine whether the ODF is designed for:
Rapid systemic absorption
Local oral action
Convenience-focused consumer use
This strategic choice directly influences polymer selection, dose loading, and packaging format [5].
Methods
Integrated Development Workflow
The one-stop ODF solution follows a structured workflow:
Concept and positioning definition
Formulation feasibility and optimization
Equipment configuration and process design
Packaging selection and stability planning
Pilot production and validation
This integrated method ensures that decisions made upstream remain compatible with downstream execution [6].
Formulation Development
Rational Formulation Design
Formulation development balances four core performance requirements:
Disintegration speed
Mechanical strength
Taste and mouthfeel
Stability
Polymer systems, plasticizers, sweeteners, and stabilizers are selected not only for laboratory performance but also for manufacturability on industrial roll-to-roll equipment [7].
Scale-Up Readiness
Formulations are optimized with drying behavior, coating uniformity, and content distribution in mind, reducing the risk of failure during scale-up [8].
Equipment and Production Line Configuration
Equipment Matching to Product Design
ODF equipment selection is driven by formulation characteristics and target capacity. Key modules include:
Precision coating systems
Multi-zone drying tunnels
Slitting and die-cutting units
Automated packaging machines
Selecting equipment in parallel with formulation development ensures process compatibility and avoids later re-engineering [9].
Modular and Scalable Design
A modular equipment strategy allows projects to move from pilot to commercial scale without fundamental process changes, supporting both OEM production and in-house manufacturing models [10].
Packaging Strategy
Protective and Functional Packaging
ODFs are highly sensitive to moisture and mechanical stress. Packaging is therefore an integral part of product design rather than a downstream add-on [11]. Common formats include sachets, stick packs, and blister wallets, chosen based on:
Shelf-life requirements
User convenience
Regulatory expectations
Branding and Compliance
Packaging design integrates branding, labeling, and compliance considerations early in development, reducing approval delays and market-entry risk [12].
Measures
Project success is evaluated using multidimensional measures [13]:
Technical feasibility (formulation and process stability)
Manufacturing robustness (yield, consistency, scalability)
Product performance (disintegration time, uniformity, taste)
Time-to-market
Cost predictability
These measures reflect both scientific and commercial readiness.
Results
Industrial application of the one-stop ODF project model demonstrates:
Reduced development cycles
Fewer formulation reworks during scale-up
Improved batch-to-batch consistency
Faster transition from concept to market
Projects developed under integrated frameworks show higher commercialization success rates compared with fragmented development approaches [14].
Discussion
The key advantage of a one-stop ODF solution lies in systems-level thinking. Unlike traditional drug development pipelines, ODF projects require early integration of engineering, materials science, and market strategy [15]. Treating formulation, equipment, and packaging as isolated steps increases risk and cost, while integrated planning enables predictable outcomes and flexible business models such as OEM, ODM, and equipment-plus-service solutions.
Conclusion
Oral Film projects achieve commercial success not through formulation excellence alone, but through coordinated development across concept positioning, formulation engineering, equipment configuration, and packaging strategy. A one-stop solution model transforms ODF development from a fragmented trial-and-error process into a structured, scalable, and market-oriented pathway. This integrated approach provides a robust foundation for accelerating innovation and commercialization in the rapidly expanding oral film sector.
References
Fu Y et al. Expert Opin Drug Deliv. 2004;1(4):673–690.
Preis M. J Pharm Pharmacol. 2013;65(2):157–170.
Dixit RP, Puthli SP. J Control Release. 2009;139(2):94–107.
Morales JO, McConville JT. Ther Deliv. 2011;2(5):637–646.
Cilurzo F et al. Eur J Pharm Biopharm. 2008;70(3):895–900.
Bala R et al. Int J Pharm Investig. 2013;3(2):67–76.
Hoffmann EM et al. Pharm Res. 2011;28(8):1914–1922.
Borges AF et al. Int J Pharm. 2015;494(1):332–339.
Preis M et al. Drug Dev Ind Pharm. 2014;40(2):152–160.
Arya A et al. Int J PharmTech Res. 2010;2(1):576–583.
Shojaei AH. J Pharm Pharm Sci. 1998;1(1):15–30.
USP <701> Disintegration Test.
USP <905> Uniformity of Dosage Units.
Keshari R, Keshari S. J Drug Deliv Ther. 2014;4(4):1–7.
Cilurzo F et al. Drug Dev Ind Pharm. 2012;38(5):588–598.