One Partner, Full Chain: The Smartest Way to Launch oral dissolving films Without Internal R&D
Author: Sihan Meng,Leyu Zhu,Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Many brands recognize the strategic value of oral dissolving films (ODFs)—for sleep, energy, immunity, pediatrics, sexual health, beauty, OTC concepts, and more—but lack in-house formulation, process, and regulatory capabilities. Building full internal R&D and manufacturing for a single new format is often economically irrational. A “one partner, full chain” model—where a specialized ODF CDMO covers ideation, formulation, pilot, scale-up, packaging, quality, and regulatory support—offers a faster, lower-risk route to market, provided it is executed with transparency, IP protection, and robust technical depth. This paper outlines how such turnkey partnerships should be structured, what capabilities to require, which metrics to track, and how to avoid common pitfalls (over-dependence, weak documentation, copy-paste formulas). Used correctly, an end-to-end ODF partner allows non-R&D brands to launch credible, differentiated products without diluting focus or control. [1–6]
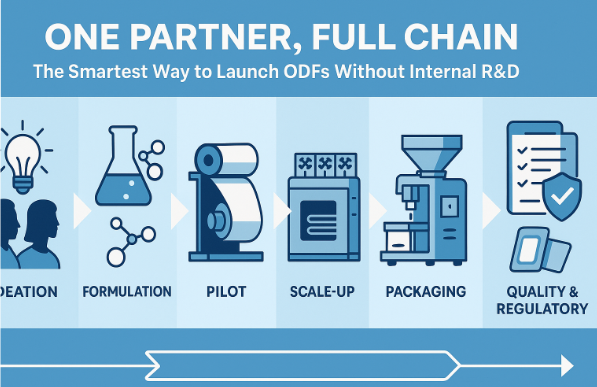
Introduction
ODFs sit at the intersection of:
Consumer expectations: convenient, portable, sugar-free/low-sugar, tech-forward formats.
Technical complexity: film matrices, taste-masking, web handling, drying, packaging, and stability.
Regulatory scrutiny: supplements vs medicines, claims, quality, data integrity.
For many marketing-led or portfolio-led companies:
Building internal ODF labs, pilot lines, and expertise:
Requires major capex + time,
Competes with core business priorities,
Is hard to justify for uncertain volume.
A specialized one-partner turnkey model can:
Collapse time-to-market,
Reduce up-front investment,
Provide instant access to proven platforms and infrastructure.
But only if:
The partner is real (true ODF platform, not just a printer),
The collaboration model protects brand/IP,
Quality, documentation, and flexibility are engineered from day one.
This paper explains how to design and evaluate that model.
Methods
We define a structured approach in four stages:
Partner Capability Mapping
Turnkey Workflow Design (idea → launch)
Risk & IP Controls
Performance Measurement
1. Partner Capability Mapping
A genuine “full chain” ODF partner should cover:
Front-End Support
Market-informed concept co-creation (sleep, kids, beauty, etc.),
Feasibility assessment (dose, actives, legal classification per region).
Formulation & Process Development
ODF matrix design,
Taste-masking strategies,
Lab and pilot-scale coating.
Scale-Up & Manufacturing
Dedicated ODF lines (multi-zone drying, web control),
Integrated slitting, die-cutting, packaging.
Quality & Regulatory
GMP / food supplement / medical device capabilities, as appropriate,
Stability, specifications, CoAs, technical dossiers.
Design & Packaging
Sachet/blister layouts,
Multi-language packs,
Export documentation support.
Absence of any of these typically forces the brand to fill gaps internally—defeating the “no R&D” goal.
2. Turnkey Workflow Design
A best-practice one-partner model follows a clear, gated workflow:
Step 1 – Concept & Target Product Profile (TPP)
Benefit area, target consumer, actives, regulatory class,
Markets, launch timing, price band, brand style.
Define:
Step 2 – Platform Matching & Feasibility
Existing ODF platforms (sleep, immune, etc.),
Regulatory constraints (dose limits, claims),
Technical feasibility (solubility, taste, stability). [1–3]
Partner maps TPP to:
Step 3 – Rapid Prototyping & Sensory
Evaluate taste, disintegration, appearance,
Adjust flavors, sweetness, branding cues.
Lab or pilot ODF samples:
Step 4 – Pilot Batches & Stability Start
Confirm manufacturability,
Start formal or accelerated stability.
Small industrially relevant batches:
Step 5 – Scale-Up & Validation
Critical process parameters,
PPQ strategy (for higher-regulated products),
Full documentation set.
Define:
Step 6 – Launch & Lifecycle
Routine production,
Ongoing stability and quality monitoring,
Variants (flavors, strengths) derived efficiently.
Throughout, the brand remains owner of positioning, claims (within law), and go-to-market; the partner owns operational excellence.
3. Risk & IP Controls
To safely rely on one partner:
Contracts
Who owns final formula,
Who owns shared improvements,
Exclusive or semi-exclusive rights where justified. [4]
NDAs/NNNs covering non-use and non-circumvention,
Clear IP clauses:
Black-Boxing
Certain premixes, branding elements, or data treated as client-owned and confidential.
For strategic clients:
Transparency & Audits
Audit rights to facilities and QMS,
Clear change-control: no unilateral changes in formula, materials, or key processes.
Portfolio Separation
Avoid partners who recycle the same concept + artwork 1:1 for many clients in same markets.
4. Performance Measurement Framework
Integrate quantitative KPIs (see Measures) into the supply and quality agreement to keep the relationship disciplined.
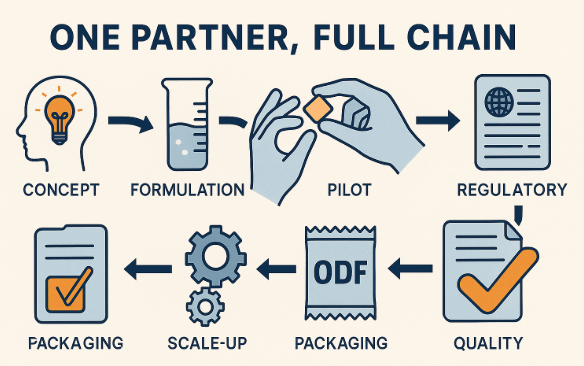
Measures
To evaluate if a one-partner full-chain model is working:
Time-to-Market
Months from project brief → shelf-ready batch.
Benchmark vs internal build or multi-vendor approach.
Technical & Quality Success
% pilot concepts that reach launch,
Batch rejection/OOS rates,
Stability pass rates at key time points.
Commercial Efficiency
COGS vs target,
Tooling/NRE transparency,
MOQ flexibility and scalability.
Compliance & Documentation
Specs, CoAs, stability reports,
Manufacturing records,
Supportive documentation for US/EU/Asia.
Availability and completeness of:
IP & Exclusivity Health
No conflicting SKUs using client’s unique formula/branding,
No disputes or red flags on misuse of data or artwork.
Relationship & Service
On-time, in-full (OTIF) delivery,
Speed of response to changes and deviations,
Satisfaction scores from brand’s technical, regulatory, and commercial teams.
Results
(Illustrative patterns from mature turnkey collaborations.)
1. Accelerated Launches
Using a capable full-chain ODF partner:
Brands frequently reduce launch timelines from 18–24 months (building internal capability) to ~6–12 months.
Early access to validated film platforms eliminates long trial-and-error cycles.
2. Lower Upfront Investment
Instead of:
Buying coating lines, dryers, packaging equipment,
Building dedicated cleanrooms and labs,
Hiring specialized formulators and engineers,
brands pay:
Clearly defined development packages,
Pilot and validation batches,
Scalable per-unit pricing.
Capex risk shifts to the partner while maintaining quality and compliance expectations.
3. Higher Technical Robustness
Turnkey specialists:
Bring proven drying, web handling, taste-masking, and packaging solutions,
Provide processes already stress-tested on multiple SKUs and markets.
As a result:
Fewer hidden scale-up surprises,
More predictable stability and sensory performance.
4. Strategic Focus for the Brand
By outsourcing the technical chain:
Brand teams focus on:
Portfolio strategy,
Channel execution,
Consumer education and digital engagement.
Internal complexity is reduced; ODFs become a plug-in format, not a structural burden.
Discussion
1. What a True “One Partner, Full Chain” Is—And Isn’t
It is:
A partner with integrated:
R&D, pilot, scale-up, QA, regulatory support, and packaging.
Able to speak both:
Technical language (CPPs, residual moisture, WVTR),
Commercial language (MOQ, lead time, landed cost, claims).
Operating under robust QMS with referenceable track record.
It is not:
A trading company outsourcing to unknown sub-suppliers,
A printer calling pre-cut films “ODFs” without process control,
A partner who refuses audits, clear contracts, or change control.
Selecting the wrong “one partner” increases risk instead of reducing it.
2. Managing Dependency Risk
Single-partner strategies raise concern about dependence. Mitigations:
Start with key SKUs, not entire portfolio,
Contractually secure:
Supply obligations,
Safety stock or dual-site options,
Technology transfer provisions in case of exit.
Once products are established, consider:
Qualifying a secondary site (even within the same group),
Securing access to critical documentation.
This preserves agility without fragmenting R&D upfront.
3. Ensuring IP & Brand Protection
To avoid becoming “just another template client”:
Clearly define what is unique:
Positioning, composition, flavor system, pack design.
Use:
NNN agreements,
IP ownership clauses,
Controls on use of your brand assets and formulations.
Monitor market for suspiciously similar products; act early if concerns arise.
4. Aligning Incentives
The model works best when:
Partner earns fair margin on both development and supply,
Brand commits to realistic volumes if KPIs are met,
Both sides share upside from:
New SKUs,
Market expansions,
Efficiency gains.
Transactional, lowest-price-only sourcing undermines trust and IP discipline
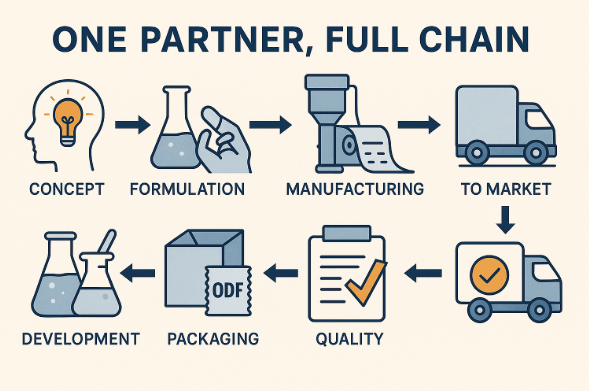
Conclusion
For companies without internal ODF R&D and manufacturing, “one partner, full chain” is often the smartest path to launch:
It compresses timelines,
Avoids heavy capex and organizational complexity,
Leverages specialized know-how in formulation, coating, drying, and packaging,
Provides a structured, document-backed route to global markets.
Success depends on choosing the right partner and structuring the relationship correctly:
Verify true ODF capabilities and QMS.
Design a clear, gated turnkey workflow from idea to launch.
Build robust IP and contract architecture.
Track performance with transparent metrics.
Maintain strategic, not blind, dependence.
Done well, this model lets brands participate in the ODF wave as serious players—not spectators—without needing to become process engineers themselves.
References
[1] Dixit RP, Puthli SP. Oral strip technology: overview and future potential. J Control Release. 2009.
[2] Preis M, Woertz C. Oromucosal films: platform for patient-centric dosage forms. J Pharm Pharmacol.
[3] ICH Q8(R2), Q9, Q10. Pharmaceutical Development; Quality Risk Management; Pharmaceutical Quality System.
[4] ISPE. Guides on technology transfer, commissioning & qualification, and outsourcing.
[5] Industry reports and case examples on CDMO-based turnkey launches in novel dosage forms (various sources).
[6] Best-practice guidance from international law and consulting firms on IP-safe manufacturing partnerships.